4 minutes agoCrime
40LENS
28 SourcesDelhi, India
Authorities Bust Fake Mounjaro Injection Racket in Gurugram, Two Arrested
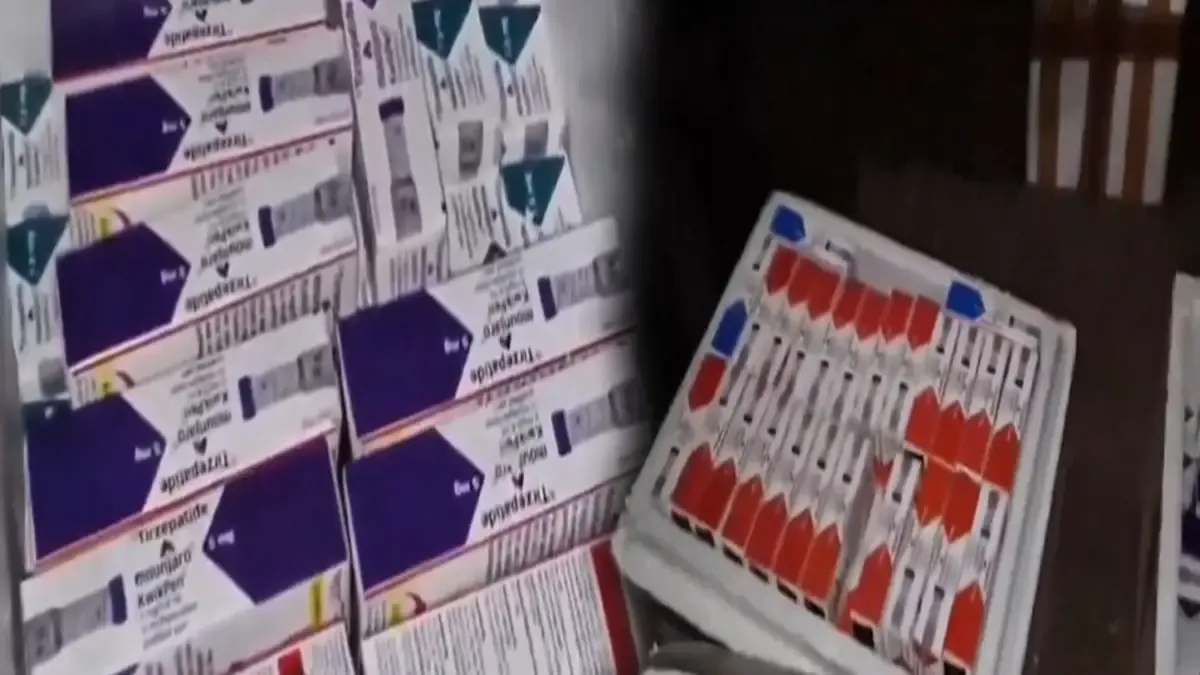
Authorities in Gurugram uncovered a counterfeit drug racket involving fake Mounjaro (tirzepatide) injections used for diabetes and weight loss. Two men were arrested after seizures of fake injections worth around Rs 56-70 lakh from vehicles and a flat in Sector 62, where the drugs were manufactured by mixing imported raw materials from China with water. The counterfeit products, sold via online portals like IndiaMART, lacked proper licensing and cold storage, raising serious health risks. Eli Lilly, the drug’s maker, confirmed the fakes and is cooperating with investigations, while a nationwide alert has been issued to detect counterfeit batches.
Political Bias
0%100%0%
Sentiment
35%
AI Analysis
Political bias across 28 sources
● Left 0%● Center 100%● Right 0%
The article group presents a largely factual and regulatory-focused perspective, emphasizing law enforcement actions and public health concerns. Sources include government officials, drug regulators, and the pharmaceutical company Eli Lilly, with no evident political framing or partisan viewpoints. Coverage centers on enforcement and safety without political commentary, reflecting a neutral stance on the issue.
Sentiment — Neutral (35/100)
The overall tone across the articles is serious and cautionary, highlighting the health risks posed by counterfeit drugs and the efforts to combat illegal manufacturing. While the coverage underscores the dangers of fake Mounjaro injections, it also notes proactive enforcement and cooperation by authorities and the drugmaker, resulting in a balanced but predominantly concerned sentiment.
How 15 sources covered this story
Each source's own headline, political lean, and sentiment — so you can see framing differences at a glance.
Coverage timeline
thetelegraph broke this story on 20 Apr, 01:00 pm. Other outlets followed.
- 1thetelegraph20 Apr, 01:00 pmIndia seizes suspected fake Mounjaro pens, says raw materials sourced from Alibaba
- 2moneycontrol20 Apr, 01:05 pmHaryana drug regulator busts fake Mounjaro racket, seizes Rs 70 lakh stock in Gurugram- Moneycontrol.com
- 3businessstandard20 Apr, 02:36 pmLilly backs regulatory action after fake Mounjaro seizure in Gurugram
- 4thetribune20 Apr, 03:29 pmFake Mounjaro racket busted in Gurugram flat; Chinese raw material mixed with water - The Tribune
- 5hindustantimes20 Apr, 05:26 pmFake Mounjaro injections manufactured in Gurugram flat; 2 arrested
- 6news1820 Apr, 05:40 pmFake Mounjaro injections manufactured in Gurugram flat; 2 arrested
- 7indiatoday20 Apr, 06:47 pmFake Mounjaro racket busted in Gurugram: Is your Mounjaro real? Here's how to check
- 8news1820 Apr, 07:59 pmFake Weight Loss Drug Racket Busted In Gurugram, Mounjaro Injections Worth Rs 56 Lakh Seized
- 9hindustantimes20 Apr, 08:52 pmFake Mounjaro injections manufactured in Gurugram flat; 2 arrested
- 10ndtv21 Apr, 04:01 amFake Weight Loss Drug Racket Busted In Gurugram, 2 Arrested
Lens Score breakdown
40/100
Public interest16/100
Coverage gap100%
Story is receiving appropriate media attention relative to public interest.
Accountability flags
TBN's analysis identified the following accountability dimensions in this story.
- public safety issue
This story involves a risk to public safety — infrastructure failure, regulatory lapse, hazardous conditions, or emergency mishandling.
Who's involved
Institutions and figures named across source coverage.
Government
Drug Control Department, GurugramGurugram Drug Control DepartmentHaryana Government
Enforcement
Police
Story context
- Category
- Crime
- Location
- Delhi, India
- Sources analysed
- 28
- Last analysed
- 21 Apr 2026
- Key entities
- CounterfeitWeight lossIndian rupeeGurgaonMedicationLakhPackaging and labelingIndiaChinaRacketeeringType 2 diabetesTirzepatide
Related Coverage
Select a news story to see related coverage from other media outlets.