2 hours agoSocial
35LENS
2 SourcesMadhya Pradesh, India
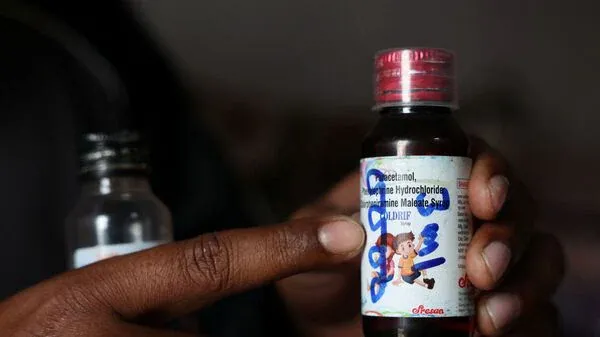
India Proposes Restrictions on Pediatric Cough Syrups Amid Safety Concerns
Following deaths linked to contaminated cough syrups in India, the health ministry plans to bar prescribing these syrups for children under two and discourage use in those under five without medical supervision. The Indian Pharmacopoeia Commission's draft National Formulary of India 2026 updates standards for high-risk ingredients prone to toxic impurities like diethylene glycol, mandating pharmacopoeial-grade solvents and rigorous batch testing to enhance safety.
Political Bias
5%93%2%
Sentiment
60%
20 stories available
View AI Analysis
Bias Analysis: The articles present a regulatory and public health perspective focused on government actions to improve drug safety. They emphasize official measures without partisan framing, reflecting a consensus on addressing contamination risks. The coverage includes government advisories and pharmaceutical standards updates, representing institutional viewpoints without political critique or opposition commentary.
Sentiment: The tone across the articles is cautious and informative, highlighting serious health risks and regulatory responses. While the reports acknowledge past fatalities linked to contaminated syrups, the emphasis is on corrective measures and safety improvements, resulting in a predominantly neutral to slightly concerned sentiment without sensationalism.
Lens Score: 35/100 — Story is receiving appropriate media attention. Public interest: 0/100. Coverage gap: 100%.
Accountability Flags: public safety issue.
Related Coverage
Select a news story to see related coverage from other media outlets.