3 hours agoSocial
35LENS
2 SourcesHyderabad, India
Hyderabad Paediatrician Faces Legal Notice from Pharma Firms over ORS Labeling Dispute
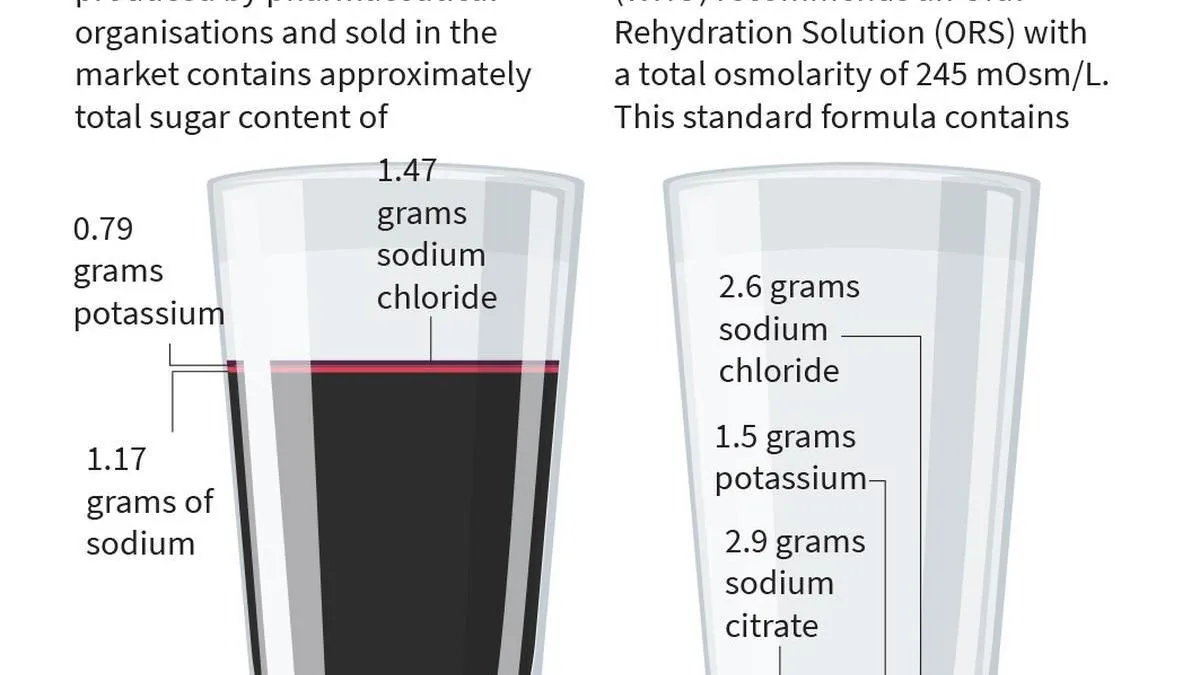
Dr. Sivaranjani Santosh, a Hyderabad paediatrician whose eight-year advocacy led the Food Safety and Standards Authority of India (FSSAI) to restrict the use of 'Oral Rehydration Salts' (ORS) labeling to formulations meeting WHO standards, is now facing legal notices from pharmaceutical companies. The firms allege she made false and defamatory remarks about their products, including ORSL and ERZL. Dr. Santosh denies these claims, citing the FSSAI order and ongoing court proceedings, with her lawyers preparing a response.
Political Bias
15%77%8%
Sentiment
42%
20 stories available
View AI Analysis
Bias Analysis: The articles present perspectives from both the paediatrician advocating for stricter ORS labeling and the pharmaceutical companies defending their products. Coverage includes regulatory actions by FSSAI and legal responses, reflecting viewpoints of government authorities, industry stakeholders, and the medical professional involved. The framing remains factual without favoring either side.
Sentiment: The tone across the articles is mixed, combining the paediatrician's successful regulatory advocacy with the legal challenges she now faces. While highlighting her efforts and the regulatory shift positively, the coverage also reports the pharmaceutical companies' allegations and legal actions, resulting in a balanced but tense sentiment.
Lens Score: 35/100 — Story is receiving appropriate media attention. Public interest: 0/100. Coverage gap: 100%.
Accountability Flags: public safety issue.
Related Coverage
Select a news story to see related coverage from other media outlets.